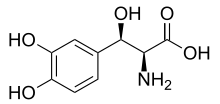
Droxidopa
 | |
| Clinical data | |
|---|---|
| Trade names | Northera |
| Routes of administration | Oral |
| ATC code | C01CA27 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 90% |
| Metabolism | Hepatic |
| Biological half-life | 1.5 hours |
| Excretion | Renal |
| Identifiers | |
| |
| Synonyms | β,3-Dihydroxytyrosine |
| CAS Number |
23651-95-8 |
| PubChem (CID) | 6989215 |
| ChemSpider | 83927 |
| UNII |
J7A92W69L7 |
| ChEBI | CHEBI:31524 |
| ChEMBL | CHEMBL2103827 |
| ECHA InfoCard | 100.215.254 |
| Chemical and physical data | |
| Formula | C9H11NO5 |
| Molar mass | 213.18734 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Droxidopa (INN; trade name Northera; also known as L-DOPS, L-threo-dihydroxyphenylserine, L-threo-DOPS and SM-5688) is a synthetic amino acid precursor which acts as a prodrug to the neurotransmitter norepinephrine (noradrenaline).[1] Unlike norepinephrine, droxidopa is capable of crossing the protective blood–brain barrier (BBB).[1]
Indications
- Neurogenic orthostatic hypotension (NOH) dopamine beta hydrolase deficiency,[2] as well as NOH associated with multiple system atrophy (MSA), familial amyloid polyneuropathy (FAP), pure autonomic failure (PAF).
- Intradialytic hypotension (IDH) or hemodialysis-induced hypotension.
- Freezing of gait in Parkinson's disease (off-label)
History
Droxidopa was developed by Sumitomo Pharmaceuticals for the treatment of hypotension, including NOH,[2] and NOH associated with various disorders such as MSA, FAP, and PD, as well as IDH. The drug has been used in Japan and some surrounding Asian areas for these indications since 1989. Following a merge with Dainippon Pharmaceuticals in 2006, Dainippon Sumitomo Pharma licensed droxidopa to Chelsea Therapeutics to develop and market it worldwide except in Japan, Korea, China, and Taiwan. In February 2014, the Food and Drug Administration approved droxidopa for the treatment of symptomatic neurogenic orthostatic hypotension.[3]
Clinical trials
Chelsea Therapeutics obtained orphan drug status (ODS) for droxidopa in the U.S. for NOH, and that of which associated with PD, PAF, and MSA. In 2014, Chelsea Therapeutics was acquired by Lundbeck along with the rights to droxidopa which was launched in the US in Sept 2014.[4]
Pharmacology
Droxidopa is a prodrug of norepinephrine used to increase the concentrations of these neurotransmitters in the body and brain.[1] It is metabolized by aromatic L-amino acid decarboxylase (AAAD), also known as DOPA decarboxylase (DDC). Patients with NOH have depleted levels of norepinephrine which leads to decreased blood pressure or hypotension upon orthostatic challenge.[5] Droxidopa works by increasing the levels of norepinephrine in the peripheral nervous system (PNS), thus enabling the body to maintain blood flow upon and while standing.
Droxidopa can also cross the blood–brain barrier (BBB) where it is converted to norepinephrine from within the brain.[1] Increased levels of norepinephrine in the central nervous system (CNS) may be beneficial to patients in a wide range of indications. Droxidopa can be coupled with a peripheral aromatic L-amino acid decarboxylase inhibitor (AAADI) or DOPA decarboxylase inhibitor (DDC) such as carbidopa (Lodosyn) to increase central norepinephrine concentrations while minimizing increases of peripheral levels.
Side effects
With over 20 years on the market, droxidopa has proven to have few side effects of which most are mild. The most common side effects reported in clinical trials include headache, dizziness nausea, hypertension and fatigue.[6][7][8][8]
References
- 1 2 3 4 Goldstein, DS (2006). "L-Dihydroxyphenylserine (L-DOPS): a norepinephrine prodrug". Cardiovasc Drug Rev. 24 (3-4): 189–203. doi:10.1111/j.1527-3466.2006.00189.x. PMID 17214596.
- 1 2 Mathias, Christopher J (2008). "L-dihydroxyphenylserine (Droxidopa) in the treatment of orthostatic hypotension". Clin Auton Res. 18 (Supplement 1): 25–29. doi:10.1007/s10286-007-1005-z.
- ↑ "FDA grants accelerated approval to NORTHERA (droxidopa) for patients with symptomatic NOH". news-medical.net. February 18, 2014.
- ↑ http://investor.lundbeck.com/releasedetail.cfm?ReleaseID=846443 http://lundbeck.com/upload/us/files/pdf/2014_Releases/NORTHERA%20Availability%20press%20release%209.2.14.pdf
- ↑ Robertson, David (2008). "The pathophysiology and diagnosis of orthostatic hypotension". Clin Auton Res. 18 (Supplement 1): 2–7. doi:10.1007/s10286-007-1004-0.
- ↑ Kaufmann, Horacio; Freeman, Roy; Biaggioni, Italo; Low, Phillip; Pedder, Simon; Hewitt, L. Arthur; Mauney, Joe; Feirtag, Michael; Mathias, Christopher J. (2014). "Droxidopa for neurogenic orthostatic hypotension: a randomized placebo-controlled Phase 3 trial". Neurology. 83 (4): 328–335. doi:10.1212/WNL.0000000000000615. PMC 4115605
 . PMID 24944260.
. PMID 24944260. - ↑ Hauser, Robert A.; Isaacson, Stuart; Lisk, Jerome P.; Hewitt, L. Arthur; Rowse, Gerry (2015). "Droxidopa for the Short-Term Treatment of Symptomatic Neurogenic Orthostatic Hypotension in Parkinson's Disease (nOH306B)". Movement Disorders. 30 (5): 646–654. doi:10.1002/mds.26086.
- 1 2 http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/203202lbl.pdf