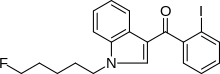
AM-694
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
335161-03-0 |
| PubChem (CID) | 9889172 |
| ChemSpider |
8064843 |
| Chemical and physical data | |
| Formula | C20H19FINO |
| Molar mass | 435.273 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
AM-694 (1-(5-fluoropentyl)-3-(2-iodobenzoyl)indole) is a drug that acts as a potent and selective agonist for the cannabinoid receptor CB1. It is used in scientific research for mapping the distribution of CB1 receptors. No public data about AM-694 metabolism is known. AM-694 has already emerged as a designer drug.
Pharmacology
AM-694 is an agonist for cannabinoid receptors. Affinities are: with a Ki of 0.08 nM at CB1 and 18x selectivity over CB2 with a Ki of 1.44 nM.[1] It is unclear what is responsible for this unusually high CB1 binding affinity, but it makes the 18F radiolabelled derivative of AM-694 useful for mapping the distribution of CB1 receptors in the body.[2]
Pharmacokinetics
AM-694 metabolism differs only slightly from that of JWH-018. AM-694 N-dealkylation produces fluoropentane instead of pentane (or plain alkanes in general). It has been speculated that the fluoropentane might function as an alkylating agent or is further metabolized into toxic fluoroacetic acid. This is not true since fluoroalkanes do not act as alkylating agents under normal conditions and uneven fluoroalkane chains metabolize into substantially less toxic fluoropropanoic acid.[3][4]
See also
- AM-679
- AM-1235
- AM-2201
- AM-2232
- AM-2233
- FUBIMINA
- JWH-018
- List of AM cannabinoids
- List of JWH cannabinoids
- THJ-2201
References
- ↑ WO patent 200128557, Makriyannis A, Deng H, "Cannabimimetic indole derivatives", granted 2001-06-07
- ↑ Willis PG, Katoch-Rouse R, Horti AG. Regioselective F-18 radiolabeling of AM694, a CB1 cannabinoid receptor ligand. Journal of Labelled Compounds and Radiopharmaceuticals 2003;46(9):799-804. doi:10.1002/jlcr.720
- ↑ Millington JE, Pattison FLM. TOXIC FLUORINE COMPOUNDS: XII. ESTERS OF ω-FLUOROALCOHOLS. Canadian Journal of Chemistry. 1956 Nov;34(11):1532-1541.
- ↑ Pattison FLM, Howell WC, Woolford RG. TOXIC FLUORINE COMPOUNDS: XIII. ω-FLUOROALKYL ETHERS. Canadian Journal of Chemistry. 1957 Feb;35(2):141-148.