Guggulsterone
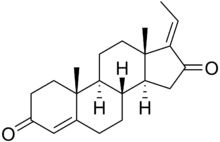 E-Guggulsterone | |
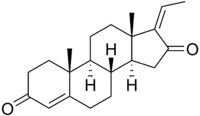 Z-Guggulsterone | |
| Names | |
|---|---|
| IUPAC name
(8R,9S,10R,13S,14S)-17-Ethylidene-10,13-dimethyl-1,2,6,7,8,9,11,12,14,15-decahydrocyclopenta[a]phenanthrene-3,16-dione | |
| Other names
Pregna-4,17-diene-3,16-dione | |
| Identifiers | |
| 95975-55-6 39025-24-6 (E) 39025-23-5 (Z) | |
| 3D model (Jmol) | (E): Interactive image (Z): Interactive image |
| ChEMBL | ChEMBL410683 |
| 2745 | |
| PubChem | 6439929 (E) 6450278 (Z) |
| UNII | A4PW148END 9B259YE66O (E) 6CST3U34GN (Z) |
| |
| Properties | |
| C21H28O2 | |
| Molar mass | 312.45 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Guggulsterone is a plant steroid found in the resin of the guggul plant, Commiphora mukul. Guggulsterone can exist as either of two stereoisomers, E-guggulsterone and Z-guggulsterone. In humans, it acts as an antagonist of the farnesoid X receptor, which was once believed to result in decreased cholesterol synthesis in the liver. Several studies have been published that indicate no overall reduction in total cholesterol occurs using various dosages of guggulsterone, and levels of low-density lipoprotein ("bad cholesterol") increased in many people.[1][2] Nevertheless, guggulsterone is an ingredient in many nutritional supplements.
Guggulsterone is a broad-spectrum ligand of steroid hormone receptors, and is known to possess the following activities:[3][4][5][6]
- Mineralocorticoid receptor antagonist (Ki = 39 nM)
- Progesterone receptor partial agonist (Ki = 201 nM)
- Glucocorticoid receptor antagonist (Ki = 224 nM)
- Androgen receptor antagonist (Ki = 240 nM)
- Estrogen receptor agonist (Ki > 5 μM; EC50 > 5 μM)
- Farnesoid X receptor antagonist (IC50 = 5–50 μM)
- Pregnane X receptor agonist (EC50 = 2.4 μM ((Z)-isomer))
Guggulsterone has been found in animal research to be orally active; it has an absolute bioavailability of 42.9% after oral administration in rats, with a half-life of around 10 hours in this species, indicating a good pharmacokinetic profile.[7]
References
- ↑ Szapary, PO; Wolfe, ML; Bloedon, LT; Cucchiara, AJ; Dermarderosian, AH; Cirigliano, MD; Rader, DJ (2003). "Guggulipid Ineffective for Lowering Cholesterol". JAMA. 290 (6): 765–772. doi:10.1001/jama.290.6.765. PMID 12915429.
- ↑ Sahni, S; Hepfinger, CA; Sauer, KA (2005). "Guggulipid Use in Hyperlipidemia". Am J Health-Syst Pharm. 62 (16): 1690–1692. doi:10.2146/ajhp040580. PMID 16085931.
- ↑ Burris, T. P. (2004). "The Hypolipidemic Natural Product Guggulsterone Is a Promiscuous Steroid Receptor Ligand". Molecular Pharmacology. 67 (3): 948–954. doi:10.1124/mol.104.007054. ISSN 0026-895X.
- ↑ Brobst, D. E. (2004). "Guggulsterone Activates Multiple Nuclear Receptors and Induces CYP3A Gene Expression through the Pregnane X Receptor". Journal of Pharmacology and Experimental Therapeutics. 310 (2): 528–535. doi:10.1124/jpet.103.064329. ISSN 0022-3565.
- ↑ Hugo Kubinyi; Gerhard Müller (6 March 2006). Chemogenomics in Drug Discovery: A Medicinal Chemistry Perspective. John Wiley & Sons. pp. 394–. ISBN 978-3-527-60402-9.
- ↑ Roger Blumenthal; JoAnne Foody; Nathan D. Wong (25 February 2011). Preventive Cardiology: A Companion to Braunwald's Heart Disease. Elsevier Health Sciences. pp. 1563–. ISBN 1-4377-3785-4.
- ↑ Verma, N.; Singh, S.K.; Gupta, R.C. (1999). "Pharmacokinetics of Guggulsterone after Intravenous and Oral Administration in Rats". Pharmacy and Pharmacology Communications. 5 (5): 349–354. doi:10.1211/146080899128734956. ISSN 1460-8081.
| ER |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| GPER |
| ||||||||||
See also: Androgenics • Glucocorticoidics • Mineralocorticoidics • Progestogenics • Steroid hormone metabolism modulators | |||||||||||
| GR |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
See also: Androgenics • Estrogenics • Mineralocorticoidics • Progestogenics • Steroid hormone metabolism modulators | |||||||||||
| MR |
| ||||||
|---|---|---|---|---|---|---|---|
See also: Androgenics • Estrogenics • Glucocorticoidics • Progestogenics • Steroid hormone metabolism modulators | |||||||
| PR |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| mPRs (PAQRs) |
| ||||||||||
See also: Androgenics • Estrogenics • Glucocorticoidics • Mineralocorticoidics • Steroid hormone metabolism modulators | |||||||||||
| CAR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ERR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| FXR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| LXR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| PPAR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| PXR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| RAR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| RXR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| SHR |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| TR |
| ||||||||||||||||||||||||||||||||||||||||||||||||