Basmisanil
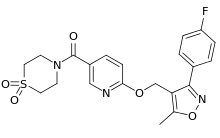 | |
| Clinical data | |
|---|---|
| ATC code | None |
| Identifiers | |
| |
| CAS Number | 1159600-41-5 |
| PubChem (CID) | 57336276 |
| ChemSpider | 34500832 |
| Chemical and physical data | |
| Formula | C21H20FN3O5S |
| Molar mass | 445.464003 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
Basmisanil (INN) (developmental code names RG-1662, RO5186582) is a highly selective inverse agonist/negative allosteric modulator of α5 subunit-containing GABAA receptors which is under development by Roche for the treatment of cognitive impairment associated with Down syndrome.[1][2][3] As of August 2015, it is in phase II clinical trials for this indication.[4]
See also
References
- ↑ Froestl W, Muhs A, Pfeifer A (2012). "Cognitive enhancers (nootropics). Part 1: drugs interacting with receptors". J. Alzheimers Dis. 32 (4): 793–887. doi:10.3233/JAD-2012-121186. PMID 22886028.
- ↑ Hurley, Dan (2013). "Investigators Silence Trisomy 21 Chromosome in Human Down Syndrome Cells". Neurology Today. 13 (17): 14–15. doi:10.1097/01.NT.0000434604.55014.fd. ISSN 1533-7006.
- ↑ Rudolph, Uwe; Jensen, Henrik Sindal; Nichol, Kathryn; Lee, Deborah; Ebert, Bjarke (2014). "Clobazam and Its Active Metabolite N-desmethylclobazam Display Significantly Greater Affinities for α2- versus α1-GABAA–Receptor Complexes". PLoS ONE. 9 (2): e88456. doi:10.1371/journal.pone.0088456. ISSN 1932-6203.
- ↑ AdisInsight. "Basmisanil". Retrieved 2015-06-10.
External links
This article is issued from Wikipedia - version of the 10/28/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.