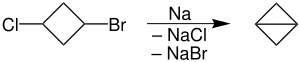Wurtz reaction
| Wurtz reaction | |
|---|---|
| Named after | Charles-Adolphe Wurtz |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | wurtz-reaction |
| RSC ontology ID | RXNO:0000074 |
The Wurtz reaction, named after Charles-Adolphe Wurtz, is a coupling reaction in organic chemistry, organometallic chemistry and recently inorganic main group polymers, whereby two alkyl halides are reacted with sodium to form a new alkane:
- 2R–X + 2Na → R–R + 2Na+X−
Other metals have also been used to effect the Wurtz coupling, among them silver, zinc, iron, activated copper, indium and a mixture of manganese and copper chloride.[1] The related reaction dealing with aryl halides is called the Wurtz-Fittig reaction.This can be explained by the formation of free radical intermediate and its subsequent disproportionation to give alkene.
Mechanism
The reaction consists of a halogen-metal exchange involving the radical species R• (in a similar fashion to the formation of a Grignard reagent and then the carbon–carbon bond formation in a nucleophilic substitution reaction.)
One electron from the metal is transferred to the halogen to produce a metal halide and an alkyl radical.
- R–X + M → R• + M+X−
The alkyl radical then accepts an electron from another metal atom to form an alkyl anion and the metal becomes cationic. This intermediate has been isolated in several cases.
- R• + M → R−M+
The nucleophilic carbon of the alkyl anion then displaces the halide in an SN2 reaction, forming a new carbon-carbon covalent bond.
- R−M+ + R–X → R–R + M+X−
Examples and reaction conditions
Due to several limitations this reaction is seldom used. For example, it is intolerant of a range of functional groups. Wurtz coupling is, however, useful in closing small, especially three-membered, rings. Bicyclobutane was prepared this way from 1-bromo-3-chlorocyclobutane in 95% yield. The reaction is conducted in refluxing dioxane, at which temperature, the sodium is liquid.[2]
Limitations
The Wurtz reaction is limited to the synthesis of symmetric alkanes. If two dissimilar alkyl halides are taken as reactants, then the product is a mixture of alkanes that is often difficult to separate as the difference in the boiling points of the products is very low in these cases, making distillation unusable. Also, since the reaction involves free radical species, a side reaction occurs to produce an alkene. This side-reaction becomes more significant when the alkyl halides are bulky at the halogen-attached carbon.Methane can not be obtained by this method.The reaction fails in case of tertiary halides.
See also
References
- ↑ March Advanced Organic Chemistry 5th edition p. 535
- ↑ Gary M. Lampman and James C. Aumiller "Bicyclo[1.1.0]butane" Organic Syntheses, 1971, volume 51, pp 55-9. doi:10.15227/orgsyn.051.0055
- Adolphe Wurtz (1855). "Sur une nouvelle classe de radicaux organiques". Annales de chimie et de physique. 44: 275–312.
- Adolphe Wurtz (1855). "Ueber eine neue Klasse organischer Radicale". Annalen der Chemie und Pharmacie. 96 (3): 364–375. doi:10.1002/jlac.18550960310.
- Organic-chemistry.org
- Organic Chemistry, by Morrison and Boyd
- Organic Chemistry, by Graham Solomons and Craig Fryhle, Wiley Publications