Quinoline Yellow SS
 | |
| Names | |
|---|---|
| Other names
Quinoline Yellow, spirit soluble; Solvent Yellow 33; C.I. Solvent Yellow 33; FD&C Yellow #11; Quinoline Yellow A; Quinoline yellow for microscopy; Yellow No. 204; C.I. 47000 | |
| Identifiers | |
| 8003-22-3 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:53700 |
| ChemSpider | 6475 |
| ECHA InfoCard | 100.029.378 |
| EC Number | 83-08-9 |
| |
| |
| Properties | |
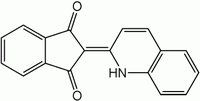
| C18H11NO2 | |
| Molar mass | 273.29 g/mol |
| Appearance | Yellow powder |
| Density | 1.34 g/cm3 |
| Melting point | 240 °C (464 °F; 513 K) |
| Insoluble | |
| Hazards | |
| R-phrases | R20/21 R33 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Quinoline Yellow SS is a bright yellow dye with green shade. It is insoluble in water, but soluble in nonpolar organic solvents. Quinoline yellow is representative of a large class of quinophthalone pigments.[1]
Synthesis and reactions
As first described in 1878, the dye is prepared by the fusion of phthalic anhydride and quinaldine. The compound exists as a mixture of two tautomers.[2] Using other anhydrides and other quinaldine derivatives other dyes in the quinophthalone family can be prepared.
When sulfonated, it converts to a water-soluble derivative, Quinoline Yellow WS.
Uses and safety
Quinoline Yellow SS is used in spirit lacquers, polystyrene, polycarbonates, polyamides, acrylic resins, and to color hydrocarbon solvents. It is also used in externally applied drugs and cosmetics. Quinoline Yellow SS is used in some yellow colored smoke formulations.
It may cause contact dermatitis. It has the appearance of a yellow powder with a melting point of 240 °C (464 °F).
References
- ↑ Volker Radtke "Quinophthalone Pigments" in High Performance Pigments (2nd Edition), Edited by Edwin B. Faulkner, Russell J. Schwartz, 2009 Wiley-VCH, Weinheim. doi:10.1002/9783527626915.ch19
- ↑ Horst Berneth "Methine Dyes and Pigments" in Ullmann's Encyclopedia of Industrial Chemistry 2008, Wiley-VCH, Weinheim. doi: 10.1002/14356007.a16_487.pub2