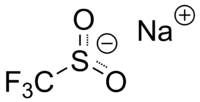
Sodium trifluoromethanesulfinate
 | |
| Names | |
|---|---|
| IUPAC name
Sodium trifluoromethanesulfinate | |
| Identifiers | |
| 2926-29-6 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 2059266 |
| ECHA InfoCard | 100.203.570 |
| PubChem | 23690734 |
| |
| |
| Properties | |
| CF3NaO2S | |
| Molar mass | 156.05 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Sodium trifluoromethanesulfinate (CF3SO2Na) is the sodium salt of trifluoromethanesulfinic acid. Together with t-butyl hydroperoxide, an oxidant, this compound was found to be a suitable reagent for introducing trifluoromethyl groups onto electron-rich aromatic compounds by Langlois; this reagent is also known as the Langlois reagent. This reaction operates via a free radical mechanism.[1]
This reagent is also able to trifluoromethylate electron-deficient aromatic compounds under biphasic conditions.[2] Zinc difluoromethanesulfinate, a related polymeric coordination complex, is able to introduce difluoromethyl groups (CHF2-) onto aromatic compounds under similar biphasic conditions as well.[3]
References
- ↑ Langlois, Bernard R.; Laurent, Eliane; Roidot, Nathalie (1991). "Trifluoromethylation of aromatic compounds with sodium trifluoromethanesulfinate under oxidative conditions". Tet. Lett. 32 (51): 7525. doi:10.1016/0040-4039(91)80524-A.
- ↑ Ji, Y.; Brueckl, T.; Baxter, R. D.; Fujiwara, Y.; Seiple, I. B.; Su, S.; Blackmond, D. G.; Baran, P. S. (2011). "Innate C-H trifluoromethylation of heterocycles". Proc. Natl. Acad. Sci. 108 (35): 14411–5. doi:10.1073/pnas.1109059108. PMC 3167544
 . PMID 21844378.
. PMID 21844378. - ↑ Fujiwara, Yuta; Dixon, Janice A.; Rodriguez, Rodrigo A.; Baxter, Ryan D.; Dixon, Darryl D.; Collins, Michael R.; Blackmond, Donna G.; Baran, Phil S. (2012). "A New Reagent for Direct Difluoromethylation". J. Am. Chem. Soc. 134 (3): 1494–7. doi:10.1021/ja211422g. PMC 3269891
 . PMID 22229949.
. PMID 22229949.
Further reading
- Simon Hadlington (2011-08-15). "Trifluoromethylation made easy". RSC Chemistry World.
This article is issued from Wikipedia - version of the 7/7/2015. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.