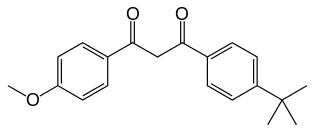
Avobenzone
 | |
 | |
| Names | |
|---|---|
| IUPAC name
1-(4-Methoxyphenyl)-3-(4-tert-butylphenyl)propane-1,3-dione | |
| Other names
butylmethoxydibenzoylmethane; 4-tert-butyl-4'-methoxydibenzoylmethane | |
| Identifiers | |
| 70356-09-1 | |
| 3D model (Jmol) | Interactive image |
| ChEMBL | ChEMBL1200522 |
| ChemSpider | 46261 |
| ECHA InfoCard | 100.067.779 |
| EC Number | 274-581-6 |
| KEGG | D03015 |
| PubChem | 51040 |
| UNII | G63QQF2NOX |
| |
| |
| Properties | |
| C20H22O3 | |
| Molar mass | 310.39 g/mol |
| Appearance | colorless crystal |
| Supplementary data page | |
| Refractive index (n), Dielectric constant (εr), etc. | |
| Thermodynamic data |
Phase behaviour solid–liquid–gas |
| UV, IR, NMR, MS | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Avobenzone (trade names Parsol 1789, Milestab 1789, Eusolex 9020, Escalol 517, Neo Heliopan 357 and others, INCI Butyl Methoxydibenzoylmethane) is an oil-soluble ingredient used in sunscreen products to absorb the full spectrum of UVA rays. It is a dibenzoylmethane derivative. Avobenzone exists in the ground state as a mixture of the enol and keto forms, favoring the chelated enol.[1] This enol form is stabilized by intramolecular hydrogen-bonding within the β-diketone.[2] Its ability to absorb ultraviolet light over a wider range of wavelengths than many other sunscreen agents has led to its use in many commercial preparations marketed as "broad spectrum" sunscreens. Avobenzone has an absorption maximum of 357 nm.[3]
History
Avobenzone was patented in 1973 and was approved in the EU in 1978. It was approved by the FDA in 1988. Its use is approved worldwide.
Avobenzone is sensitive to the properties of the solvent, being relatively stable in polar protic solvents and unstable in nonpolar environments. Also, when it is irradiated with UVA light, it generates a triplet excited state in the keto form which can either cause the avobenzone to degrade or it can transfer energy to biological targets and cause deleterious effects.[1]
Avobenzone has been shown to degrade significantly in light, resulting in less protection over time.[4][5][6] The UV-A light in a day of sunlight in a temperate climate is sufficient to break down most of the compound. Data presented to the Food and Drug Administration by the Cosmetic, Toiletry and Fragrance Association indicates a -36% change in avobenzone's UV absorbance following one hour of exposure to sunlight.[7] This degradation can be reduced by using a photostabilizer, like octocrylene. Other photostabilizers include:
- 4-Methylbenzylidene camphor (USAN Enzacamene)
- Tinosorb S (USAN Bemotrizinol, INCI Bis-Ethylhexyloxyphenol Methoxyphenyl Triazine)
- Tinosorb M (USAN Bisoctrizole, INCI Methylene Bis-Benzotriazolyl Tetramethylbutylphenol)
- Butyloctyl Salicylate (Tradename HallBrite BHB - )
- Hexadecyl Benzoate
- Butyloctyl Benzoate
- HallBrite PSF (INCI UNDECYLCRYLENE DIMETHICONE)[8]
- Mexoryl SX (USAN Ecamsule, INCI Terephthalylidene Dicamphor Sulfonic Acid)
- Corapan TQ (INCI Diethylhexyl 2,6-Naphthalate)[9]
- Parsol SLX (INCI Polysilicone-15)[10]
- Oxynex ST (INCI Diethylhexyl Syringylidene Malonate[11]
- Polycrylene (INCI Polyester-8)[12]
- SolaStay S1 (INCI Ethylhexyl Methoxycrylene)[13]
Complexing avobenzone with cyclodextrins may also increase its photostability.[14] Formulations of avobenzone with hydroxypropyl-beta-cyclodextrin have shown significant reduction in photo-induced degradation, as well as increased transdermal penetration of the UV absorber.[15]
According to some studies, "the most effective sunscreens contain avobenzone and titanium dioxide."[16][17] Avobenzone can degrade faster in light in combination with mineral UV absorbers like zinc oxide and titanium dioxide, though with the right coating of the mineral particles this reaction can be reduced.[18] A manganese doped titanium dioxide may be better than undoped titanium dioxide to improve avobenzone's stability.[19]
Avobenzone reacts with minerals to form colored complexes. Manufacturers of avobenzone, like DSM recommend to include a chelator to prevent this from happening. They also recommend to avoid the inclusion of iron and ferric salts, heavy metals, formaldehyde donors and PABA and PABA esters.
The makers of Coppertone advise that avobenzone binds iron and can cause staining of clothes washed in iron-rich water.
Avobenzone also reacts with boron trifluoride to form a stable crystalline complex that is highly fluorescent under UV irradiation. More interestingly, the emission color of the crystals relies on the molecular packing of the boron avobenzone complex. The photoluminescence may also be altered by mechanical force in the solid state, resulting in a phenomenon called "mechanochromic luminescence". The altered emission color recovers itself slowly at room temperature or more swiftly at higher temperatures.[20]
See also
Notes
- 1 2 Paris C, Lhiaubet-Vallet V, Jimenez O, Trullas C, Miranda M (January–February 2009). "A Blocked Diketo Form of Avobenzone: Photostability, Photosensitizing Properties and Triplet Quenching by a Triazine-derived UVB-filter". Photochemistry and Photobiology. 85 (1): 178–184. doi:10.1111/j.1751-1097.2008.00414.x. PMID 18673327.
- ↑ Zawadiak J, Mrzyczek M (October 2012). "Influence Of Substituent On UV Absorption And Keto–Enol Tautomerism Equilibrium Of Dibenzoylmethane Derivatives". Spectrochim Acta A Mol Biomol Spectrosc. 96: 815–9. doi:10.1016/j.saa.2012.07.109. PMID 22925908.
- ↑ Vielhaber G, Grether-Beck S, Koch O, Johncock W, Krutmann J (March 2006). "Sunscreens with an absorption maximum of > or =360 nm provide optimal protection against UVA1-induced expression of matrix metalloproteinase-1, interleukin-1, and interleukin-6 in human dermal fibroblasts". Photochem Photobiol Sci. 5 (3): 275–82. doi:10.1039/b516702g. PMID 16520862.
- ↑ Chatelain E, Gabard B (September 2001). "Photostabilization of Butyl methoxydibenzoylmethane (Avobenzone) and Ethylhexyl methoxycinnamate by Bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S), a new UV broadband filter". Photochem Photobiol. 74 (3): 401–6. doi:10.1562/0031-8655(2001)074<0401:POBMAA>2.0.CO;2. PMID 11594052.
- ↑
 Tarras-Wahlberg N, Stenhagen G, Larko O, Rosen A, Wennberg AM, Wennerstrom O (October 1999). "Changes in ultraviolet absorption of sunscreens after ultraviolet irradiation". J Invest Dermatol. 113 (4): 547–53. doi:10.1046/j.1523-1747.1999.00721.x. PMID 10504439.
Tarras-Wahlberg N, Stenhagen G, Larko O, Rosen A, Wennberg AM, Wennerstrom O (October 1999). "Changes in ultraviolet absorption of sunscreens after ultraviolet irradiation". J Invest Dermatol. 113 (4): 547–53. doi:10.1046/j.1523-1747.1999.00721.x. PMID 10504439. - ↑ Wetz F, Routaboul C, Denis A, Rico-Lattes I (Mar–Apr 2005). "A new long-chain UV absorber derived from 4-tert-butyl-4'-methoxydibenzoylmethane: absorbance stability under solar irradiation". J Cosmet Sci. 56 (2): 135–48. doi:10.1562/2004-03-09-ra-106. PMID 15870853.
- ↑ CTFA letter re: Tentative Final Monograph for OTC Sunscreen
- ↑ http://www.hallstar.com/pis.php?product=1A054
- ↑ Bonda C.; Steinberg D. C. (2000). "A new photostabilizer for full spectrum sunscreens". Cosmet. Toiletries. 115 (6): 37–45.
- ↑ http://www.dsm.com/en_US/downloads/dnp/Parsol_SLX_Skin.pdf
- ↑ Chaudhuri RK, Lascu Z, Puccetti G, Deshpande AA, Paknikar SK (May–Jun 2006). "Design of a photostabilizer having built-in antioxidant functionality and its utility in obtaining broad-spectrum sunscreen formulations". Photochem Photobiol. 82 (3): 823–8. doi:10.1562/2005-07-15-RA-612. PMID 16492073.
- ↑ http://www.hallstar.com/techdocs/Polycrylene&CorapanTQAvobenzoneStabilization.pdf
- ↑ "Product Information Sheet: SolaStay S1". The HallStar Company. Retrieved 16 February 2010.
- ↑ Scalia S, Simeoni S, Barbieri A, Sostero S (November 2002). "Influence of hydroxypropyl-beta-cyclodextrin on photo-induced free radical production by the sunscreen agent, butyl-methoxydibenzoylmethane". J Pharm Pharmacol. 54 (11): 1553–8. doi:10.1211/002235702207. PMID 12495559.
- ↑ Yang J, Wiley C, Godwin D, Felton L (June 2008). "Influence of hydroxypropyl-β-cyclodextrin on transdermal penetration and photostability of avobenzone". Eur J Pharm Biopharm. 69 (2): 605–612. doi:10.1016/j.ejpb.2007.12.015. PMID 18226883.
- ↑ Warwick L. Morison, M.D. (March 11, 2004). "Photosensitivity". The New England Journal of Medicine. 350 (11): 1111–1117. doi:10.1056/NEJMcp022558. PMID 15014184.
- ↑ "Sunscreen Drug Products for Over-the-Counter use; Marketing Status of Products Containing Avobenzone; Enforcement Policy" (PDF). US Food and Drug Administration. 1997-04-30. p. 23354. Archived (PDF) from the original on 2007-02-26. Retrieved 2007-06-03.
- ↑ Stability Study of Avobenzone with Inorganic Sunscreens, Kobo Products Poster, 2001, Online version Archived May 7, 2007, at the Wayback Machine.
- ↑ Wakefield G, Lipscomb S, Holland E, Knowland J (July 2004). "The effects of manganese doping on UVA absorption and free radical generation of micronised titanium dioxide and its consequences for the photostability of UVA absorbing organic sunscreen components". Photochem Photobiol Sci. 3 (7): 648–52. doi:10.1039/b403697b. PMID 15238999.
- ↑ Zhang G; Lu J; Sabat M; Fraser, CL (February 2010). "Polymorphism and Reversible Mechanochromic Luminescence for Solid-State Difluoroboron Avobenzone". J. Am. Soc. Chem. 132 (7): 2160–2162. doi:10.1021/ja9097719. PMID 20108897.