Oleyl alcohol
 | |
| Names | |
|---|---|
| IUPAC name
(Z)-Octadec-9-en-1-ol | |
| Other names
Octadecenol cis-9-Octadecen-1-ol | |
| Identifiers | |
| 143-28-2 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:73504 |
| ChemSpider | 4447562 |
| ECHA InfoCard | 100.005.089 |
| KEGG | D05245 |
| PubChem | 5284499 |
| UNII | 172F2WN8DV |
| |
| |
| Properties | |
| C18H36O | |
| Molar mass | 268.478 g/mol |
| Density | 0.845-0.855 g/cm3 |
| Melting point | 13 to 19 °C (55 to 66 °F; 286 to 292 K) |
| Boiling point | 330 to 360 °C (626 to 680 °F; 603 to 633 K) |
| Insoluble | |
| Hazards | |
| NFPA 704 | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
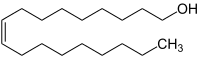
Oleyl alcohol, octadecenol, or cis-9-octadecen-1-ol, is an unsaturated fatty alcohol with the chemical formula C18H36O or CH3(CH2)7-CH=CH-(CH2)8OH.
It can be produced by the hydrogenation of oleic acid esters;[1] which can be obtained naturally from beef fat, fish oil and in particular olive oil (from which it gains its name).
It has uses as a nonionic surfactant, emulsifier, emollient and thickener in skin creams, lotions and many other cosmetic products including shampoos and hair conditioners. It has also been investigated as a carrier for delivering medications through the skin or mucus membranes; particularly the lungs.[2]
See also
- Oleic acid - the corresponding fatty acid
- Oleylamine - the corresponding amine
- Oleamide - the corresponding amide
References
- ↑ Kreutzer, Udo R. (February 1984). "Manufacture of fatty alcohols based on natural fats and oils". Journal of the American Oil Chemists' Society. 61 (2): 343–348. doi:10.1007/BF02678792.
- ↑ Hussain, Alamdar; Arnold, John J.; Khan, Mansoor A.; Ahsan, Fakhrul (January 2004). "Absorption enhancers in pulmonary protein delivery". Journal of Controlled Release. 94 (1): 15–24. doi:10.1016/j.jconrel.2003.10.001. PMID 14684268.
This article is issued from Wikipedia - version of the 9/27/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.
