Neuraminidase inhibitor
Neuraminidase inhibitors (NAIs) are a class of drugs which block the neuraminidase enzyme. They are commonly used as antiviral drugs because they block the function of viral neuraminidases of the influenza virus, by preventing its reproduction by budding from the host cell. Oseltamivir (Tamiflu) a prodrug, Zanamivir (Relenza), Laninamivir (Inavir), and Peramivir belong to this class. Unlike the M2 inhibitors, which work only against the influenza A, neuraminidase inhibitors act against both influenza A and influenza B.[1][2][3][4] The neuraminidase inhibitors oseltamivir and zanamivir were approved in the US and Europe for treatment and prevention of influenza A and B. Peramivir acts by strongly binding to the neuraminidase of the influenza viruses and inhibits activation of neuraminidase much longer than Oseltamivir or Zanamivir.[5] However, Laninamivir in the cells is slowly released into the respiratory tract, resulting in long-lasting anti-influenza virus activity. Thus the mechanism of the long-lasting activity of laninamivir is basically different from that of Peramivir.[6]
The efficacy was highly debated in recent years.[7][8][9] However, after the pandemic caused by H1N1 in 2009, the effectiveness of early treatment with neuraminidase inhibitors in reducing serious cases and deaths was reported in various countries.[10][11][12][13]
In countries where treatment of influenza like illness are done using neuraminidase inhibitors on a national level, statistical reports show a low fatality record for symptomatic illness because of the universal implementation of early treatment using NAIs.[14] Although oseltamivir is widely used in these countries there has been no outbreaks caused by oseltamivir-resistant viruses and also no serious illness caused by oseltamivir-resistant viruses has ever been reported.[14] The United States Centers for Disease Control continues to recommend the use of oseltamavir treatment for people at high risk for complications and the elderly and those at lower risk who present within 48 hours of first symptoms of infection.[15]
Common side effects include nausea and vomiting. The abnormal behaviours of children after taking oseltamivir that have been reported may be an extension of delirium or hallucinations caused by influenza.[14] It occurs in the early stages of the illness, such as within 48 hours after onset of the illness. Therefore, children with influenza are advised to be observed by their parents until 48 hours after the onset of the influenza illness, regardless of whether the child is treated with NAIs.[14]
Specific neuraminidase inhibitors
- Laninamivir
- Oseltamivir (Tamiflu)
- Peramivir
- Zanamivir (Relenza)
Structures of the Viral neuraminidase inhibitors in use
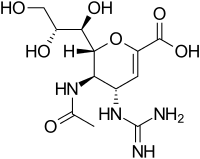 |
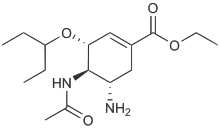 |
 |
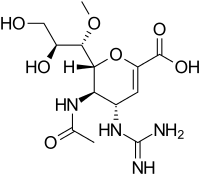 |
| Zanamivir | Oseltamivir | Peramivir | Laninamivir |
Natural products
- Cyanidin-3-sambubioside (Extracted from black elderberry)[16]
- Coptisine
- Berberine[17]
See also
References
- ↑ Smith, B. J.; McKimm-Breshkin, J. L.; McDonald, M.; Fernley, R. T.; Varghese, J. N.; Colman, P. M. (2002). "Structural Studies of the Resistance of Influenza Virus Neuramindase to Inhibitors". Journal of Medicinal Chemistry. 45 (11): 2207–12. doi:10.1021/jm010528u. PMID 12014958.
- ↑ Gubareva, Larisa V (2004). "Molecular mechanisms of influenza virus resistance to neuraminidase inhibitors". Virus Research. 103 (1-2): 199–203. doi:10.1016/j.virusres.2004.02.034. PMID 15163510.
- ↑ This flash animation shows the mode of action of oseltamivir (Tamiflu). pharmasquare.org
- ↑ Replication of influenza virus. mvm.ed.ac.uk
- ↑ Bantia, Shanta; Arnold, C. Shane; Parker, Cynthia D.; Upshaw, Ramanda; Chand, Pooran (1 January 2006). "Anti-influenza virus activity of peramivir in mice with single intramuscular injection". Antiviral Research. 69 (1): 39–45. doi:10.1016/j.antiviral.2005.10.002. ISSN 0166-3542. PMID 16325932.
- ↑ Ishizuka, Hitoshi; Yoshiba, Satoshi; Okabe, Hiromi; Yoshihara, Kazutaka (1 November 2010). "Clinical pharmacokinetics of laninamivir, a novel long-acting neuraminidase inhibitor, after single and multiple inhaled doses of its prodrug, CS-8958, in healthy male volunteers". Journal of Clinical Pharmacology. 50 (11): 1319–1329. doi:10.1177/0091270009356297. ISSN 1552-4604. PMID 20145259.
- ↑ Jefferson, Tom; Jones, Mark; Doshi, Peter; Spencer, Elizabeth A.; Onakpoya, Igho; Heneghan, Carl J. (9 April 2014). "Oseltamivir for influenza in adults and children: systematic review of clinical study reports and summary of regulatory comments". BMJ. 348: g2545. doi:10.1136/bmj.g2545. ISSN 1756-1833. PMC 3981975
 . PMID 24811411.
. PMID 24811411. - ↑ Hsu, Jonathan; Santesso, Nancy; Mustafa, Reem; Brozek, Jan; Chen, Yao Long; Hopkins, Jessica P.; Cheung, Adrienne; Hovhannisyan, Gayane; Ivanova, Liudmila; Flottorp, Signe A.; Saeterdal, Ingvil; Wong, Arthur D.; Tian, Jinhui; Uyeki, Timothy M.; Akl, Elie A.; Alonso-Coello, Pablo; Smaill, Fiona; Schünemann, Holger J. (3 April 2012). "Antivirals for treatment of influenza: a systematic review and meta-analysis of observational studies". Annals of Internal Medicine. 156 (7): 512–524. doi:10.7326/0003-4819-156-7-201204030-00411. ISSN 1539-3704. PMID 22371849.
- ↑ Heneghan, Carl J.; Onakpoya, Igho; Thompson, Matthew; Spencer, Elizabeth A.; Jones, Mark; Jefferson, Tom (9 April 2014). "Zanamivir for influenza in adults and children: systematic review of clinical study reports and summary of regulatory comments". BMJ. 348: g2547. doi:10.1136/bmj.g2547. ISSN 1756-1833. PMC 3981976
 . PMID 24811412.
. PMID 24811412. - ↑ Louie, Janice K.; Acosta, Meileen; Jamieson, Denise J.; Honein, Margaret A. "Severe 2009 H1N1 Influenza in Pregnant and Postpartum Women in California". New England Journal of Medicine. 362 (1): 27–35. doi:10.1056/nejmoa0910444.
- ↑ Jain, Seema; Kamimoto, Laurie; Bramley, Anna M.; Schmitz, Ann M.; Benoit, Stephen R.; Louie, Janice; Sugerman, David E.; Druckenmiller, Jean K.; Ritger, Kathleen A.; Chugh, Rashmi; Jasuja, Supriya; Deutscher, Meredith; Chen, Sanny; Walker, John D.; Duchin, Jeffrey S.; Lett, Susan; Soliva, Susan; Wells, Eden V.; Swerdlow, David; Uyeki, Timothy M.; Fiore, Anthony E.; Olsen, Sonja J.; Fry, Alicia M.; Bridges, Carolyn B.; Finelli, Lyn (12 November 2009). "Hospitalized Patients with 2009 H1N1 Influenza in the United States, April–June 2009". New England Journal of Medicine. 361 (20): 1935–1944. doi:10.1056/NEJMoa0906695. ISSN 0028-4793. PMID 19815859.
- ↑ Domínguez-Cherit, Guillermo; Lapinsky, Stephen E.; Macias, Alejandro E.; Pinto, Ruxandra; Espinosa-Perez, Lourdes; de la Torre, Alethse; Poblano-Morales, Manuel; Baltazar-Torres, Jose A.; Bautista, Edgar; Martinez, Abril; Martinez, Marco A.; Rivero, Eduardo; Valdez, Rafael; Ruiz-Palacios, Guillermo; Hernández, Martín; Stewart, Thomas E.; Fowler, Robert A. (4 November 2009). "Critically Ill patients with 2009 influenza A(H1N1) in Mexico". JAMA. 302 (17): 1880–1887. doi:10.1001/jama.2009.1536. ISSN 1538-3598. PMID 19822626.
- ↑ Zarychanski, R.; Stuart, T. L.; Kumar, A.; Doucette, S.; Elliott, L.; Kettner, J.; Plummer, F. (2010). "Correlates of severe disease in patients with 2009 pandemic influenza (H1N1) virus infection". Canadian Medical Association Journal. 182 (3): 257–264. doi:10.1503/cmaj.091884. ISSN 0820-3946.
- 1 2 3 4 Sugaya, Norio (1 October 2011). "Widespread use of neuraminidase inhibitors in Japan". Journal of Infection and Chemotherapy: Official Journal of the Japan Society of Chemotherapy. 17 (5): 595–601. doi:10.1007/s10156-011-0288-0. ISSN 1437-7780. PMID 21850418.
- ↑ "CDC Online Newsroom - "Have You Heard?" Archive: 2014 - Influenza A Variant Virus". cdc.gov.
- ↑ Swaminathan K, Dyason JC, Maggioni A, von Itzstein M, Downard KM (2013). "Binding of a natural anthocyanin inhibitor to influenza neuraminidase by mass spectrometry". Anal Bioanal Chem. 405 (20): 6563–72. doi:10.1007/s00216-013-7068-x. PMID 23748498.
- ↑ Kim, Jang Hoon; Ryu, Young Bae; Lee, Woo Song; Kim, Young Ho (2014-11-01). "Neuraminidase inhibitory activities of quaternary isoquinoline alkaloids from Corydalis turtschaninovii rhizome". Bioorganic & Medicinal Chemistry. 22 (21): 6047–6052. doi:10.1016/j.bmc.2014.09.004. ISSN 1464-3391. PMID 25277281.