Potassium bis(trimethylsilyl)amide
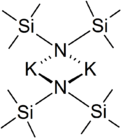 | |
amide_unsolvated_from_crystal.png) | |
| Names | |
|---|---|
| Preferred IUPAC name
Potassium bis(trimethylsilyl)amide | |
| Other names
Potassium hexamethyldisilazide | |
| Identifiers | |
| 40949-94-8 | |
| 3D model (Jmol) | Interactive image |
| Abbreviations | KHMDS |
| ChemSpider | 21171158 |
| ECHA InfoCard | 100.115.605 |
| PubChem | 3251421 |
| UN number | 3263 |
| |
| |
| Properties | |
| KSi 2C 6NH 18 | |
| Molar mass | 199.4831 g mol−1 |
| Appearance | White, opaque crystals |
| Reacts | |
| Hazards | |
| GHS pictograms |  |
| GHS signal word | DANGER |
| H314[1] | |
| P280, P305+351+338, P310[1] | |
| EU classification (DSD) |
|
| R-phrases | R14, R34[1] |
| S-phrases | S26, S36/37/39, S43, S45[1] |
| Related compounds | |
| Other cations |
Lithium bis(trimethylsilyl)amide |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Potassium bis(trimethylsilyl)amide (commonly abbreviated as KHMDS, Potassium(K) HexaMethylDiSilazide) is the chemical compound with the formula ((CH3)3Si)2NK. It is a strong, non-nucleophilic base with an approximate pKa of 26 (compare to lithium diisopropylamide, at 36).
Structure
In the solid state, the unsolvated compound is dimeric, with two potassium and two nitrogen atoms forming a square. This compound is soluble in hydrocarbon solvents, and conducts electricity poorly in solution and in the melt. This is attributed to very strong ion-pairing.[2]
See also
References
- 1 2 3 4 5 Potassium bis(trimethylsilyl)amide, Sigma-Aldrich
- ↑ Tesh, Kris F.; Hanusa, Timothy P.; Huffman, John C. (1990). "Ion pairing in [bis(trimethylsilyl)amido]potassium: The x-ray crystal structure of unsolvated [KN(SiMe3)2]2". Inorg. Chem. 29 (8): 1584–1586. doi:10.1021/ic00333a029.
This article is issued from Wikipedia - version of the 7/20/2015. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.