Calcium monophosphide
 | |
| Names | |
|---|---|
| Other names
Calcium phosphide | |
| Identifiers | |
| 39373-03-0 | |
| ECHA InfoCard | 100.049.465 |
| Properties | |
| CaP (Ca2P2) | |
| Appearance | black solid |
| decomposes | |
| Hazards | |
| R-phrases | R15/29 R28 R50 |
| S-phrases | (S1/2) S22 S43 S45 S61 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Calcium monophosphide is the inorganic compound with the formula CaP. The term "calcium phosphide" also describes the composition Ca3P2, which is also called calcium phosphide. CaP and Ca3P2 are completely different materials. CaP is black, Ca3P2 is red-brown. The monophosphide decomposes to Ca3P2 at about 600 °C.
- 3 CaP → Ca3P2 + 1/4 P4
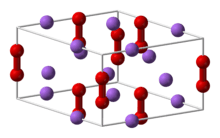
Structure and properties
The structures of CaP and sodium peroxide (Na2O2) are very similar.[1] The solid is described as a salt: (Ca2+)2P24−, or Ca2P2. Since the bonding is ionic, the diphosphide centers carry negative charge and are easily protonated. Upon hydrolysis this material releases diphosphine (P2H4):[2]
- Ca2P2 + 4 H2O → 2 Ca(OH)2 + P2H4
The hydrolyses of CaP and calcium carbide (CaC2) are similar, except that diphosphine spontaneously ignites in air. Thus, CaP must be protected from air.
References
- ↑ Iandelli, A. and Franceschi, E., "On the crystal structure of the compounds CaP, SrP, CaAs, SrAs and EuAs", Journal of the Less Common Metals, 1973, volume 30, pp. 211-216. doi:10.1016/0022-5088(73)90107-0
- ↑ Marianne Baudler, Klaus Glinka (1993). "Monocyclic and polycyclic phosphines". Chem. Rev. 93 (4): 1623–1667. doi:10.1021/cr00020a010.