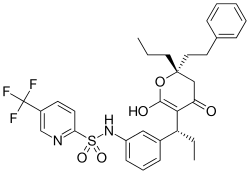
Tipranavir
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Aptivus |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a606009 |
| License data |
|
| Pregnancy category | |
| Routes of administration | Oral |
| ATC code | J05AE09 (WHO) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | N/D |
| Protein binding | 99.9% |
| Metabolism | Hepatic |
| Biological half-life | 4.8–6 hours |
| Excretion | Feces (82.3%), urine (4.4%) |
| Identifiers | |
| |
| CAS Number |
174484-41-4 |
| PubChem (CID) | 65027 |
| DrugBank |
DB00932 |
| ChemSpider |
10482313 |
| UNII |
ZZT404XD09 |
| ChEMBL |
CHEMBL183041 |
| NIAID ChemDB | 032941 |
| PDB ligand ID | TPV (PDBe, RCSB PDB) |
| ECHA InfoCard | 100.158.066 |
| Chemical and physical data | |
| Formula | C31H33F3N2O5S |
| Molar mass | 602.66 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Tipranavir, or tipranavir disodium, is a nonpeptidic protease inhibitor (PI) manufactured by Boehringer-Ingelheim under the trade name Aptivus. It is administered with ritonavir in combination therapy to treat HIV infection.
Tipranavir has the ability to inhibit the replication of viruses that are resistant to other protease inhibitors and it recommended for patients who are resistant to other treatments. Resistance to tipranavir itself seems to require multiple mutations.[1]
Tipranavir was approved by the Food and Drug Administration (FDA) on June 22, 2005, and was approved for pediatric use on June 24, 2008.[2] Like lopinavir and atazanavir, it is very potent and is effective in salvage therapy for patients with some drug resistance. However, side effects of tipranavir may be more severe than those of other anti-retrovirals. Some side effects include intracranial hemorrhage, hepatitis, hyperglycemia and diabetes mellitus. The drug has also been shown to cause increases in total cholesterol and triglycerides.[3]
References
- ↑ Doyon L, Tremblay S, Bourgon L, Wardrop E, Cordingley M (2005). "Selection and characterization of HIV-1 showing reduced susceptibility to the non-peptidic protease inhibitor tipranavir.". Antiviral Res. 68 (1): 27–35. doi:10.1016/j.antiviral.2005.07.003. PMID 16122817.
- ↑ "New Aptivus (tipranavir) Oral Solution Approved for Treatment-Experienced Pediatric and Adolescent HIV Patients" (Press release). Boehringer Ingelheim. 2008-06-24. Retrieved 2008-09-02.
- ↑ "Aptivus® (tipranavir) capsules 250 mg, oral solution 100 mg/mL, Prescribing Information", Boehringer Inhelheim, 2012. Revised April 2012.