1,5-Cyclooctadiene
 | |
 | |
| Names | |
|---|---|
| Systematic IUPAC name
Cycloocta-1,5-diene[1] | |
| Identifiers | |
| 111-78-4 1552-12-1 (Z,Z) 5259-71-2 (Z,E) 17612-50-9 (E,E) | |
| 3D model (Jmol) | Interactive image |
| Abbreviations | 1,5-COD |
| 2036542 1209288 (Z,Z) | |
| ChemSpider | 7843 74815 (Z,Z) 18520443 (Z,E) 19971660 (E,E) |
| ECHA InfoCard | 100.003.552 |
| EC Number | 203-907-1 |
| MeSH | 1,5-cyclooctadiene |
| PubChem | 8135 82916 (Z,Z) 5364364 (Z,E) 5702534 (E,E) |
| RTECS number | GX9560000 GX9620000 (Z,Z) |
| UN number | 2520 |
| |
| |
| Properties | |
| C8H12 | |
| Molar mass | 108.18 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 0.882 g/mL |
| Melting point | −69 °C; −92 °F; 204 K |
| Boiling point | 150 °C; 302 °F; 423 K |
| Vapor pressure | 910 Pa |
| Refractive index (nD) |
1.493 |
| Thermochemistry | |
| 198.9 J K−1 mol−1 | |
| Std molar entropy (S |
250.0 J K−1 mol−1 |
| Std enthalpy of formation (ΔfH |
21–27 kJ mol−1 |
| Std enthalpy of combustion (ΔcH |
−4.890 – −4.884 MJ mol−1 |
| Hazards | |
| GHS pictograms |   |
| GHS signal word | DANGER |
| H226, H304, H315, H317, H319, H334 | |
| P261, P280, P301+310, P305+351+338, P331, P342+311 | |
| EU classification (DSD) |
|
| R-phrases | R10, R36/38, R42/43, R65 |
| S-phrases | S23, S26, S36/37, S62 |
| Flash point | 32 to 38 °C (90 to 100 °F; 305 to 311 K) |
| 222 °C (432 °F; 495 K) | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
1,5-Cyclooctadiene is the organic compound with the chemical formula C8H12. Generally abbreviated COD, this diene is a useful precursor to other organic compounds and serves as a ligand in organometallic chemistry. It is a colorless liquid with a strong odor.[2][3] 1,5-Cyclooctadiene can be prepared by dimerization of butadiene in the presence of a nickel catalyst, a coproduct being vinylcyclohexene. Approximately 10,000 tons were produced in 2005.[4]
Organic reactions
COD reacts with borane to give 9-borabicyclo[3.3.1]nonane,[5] commonly known as 9-BBN, a reagent in organic chemistry used in hydroborations:
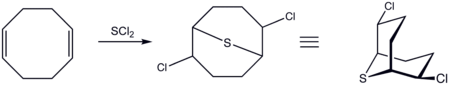
COD adds SCl2 (or similar reagents) to give 2,6-dichloro-9-thiabicyclo[3.3.1]nonane:[6][7]
The resulting dichloride can be further modified as the diazide or dicyano derivative in a nucleophilic substitution aided by anchimeric assistance.
Metal complexes
- Selected metal 1,5-COD complexes.
-

The complex Rh2(COD)2Cl2.
-
(C8H13).png)
Co(1,5-cyclooctadiene)(cyclooctenyl).
1,5-COD binds to low-valent metals via both alkene groups. Metal-COD complexes are attractive because they are sufficiently stable to be isolated, often being more robust than related ethylene complexes. The stability of COD complexes is attributable to the chelate effect. The COD ligands are easily displaced by other ligands, such as phosphines.
Ni(COD)2 is prepared by reduction of anhydrous nickel acetylacetonate in the presence of the ligand, using triethylaluminium [8]
- 1⁄3 [Ni(C5H7O2)2]3 + 2 COD + 2 Al(C2H5)3 → Ni(COD)2 + 2 Al(C2H5)2(C5H7O2) + C2H4 + C2H6
The related Pt(COD)2 is prepared by a more circuitous route involving the dilithium cyclooctatetraene:[9]
- Li2C8H8 + PtCl2(COD) + 3 C7H10 → [Pt(C7H10)3] + 2 LiCl + C8H8 + C8H12
- Pt(C7H10)3 + 2 COD → Pt(COD)2 + 3 C7H10
Extensive work has been reported on complexes of COD, much of which has been described in volumes 25, 26, and 28 of Inorganic Syntheses. The platinum complex is a precursor to a 16-electron complex of ethylene:
- Pt(COD)2 + 3 C2H4 → Pt(C2H4)3 + 2 COD
COD complexes are useful as starting materials; one noteworthy example is the reaction:
- Ni(COD)2 + 4 CO → Ni(CO)4 + 2 COD
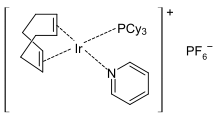
The product Ni(CO)4 is highly toxic, thus it is advantageous to generate it in the reaction vessel upon demand. Other low-valent metal complexes of COD include cyclooctadiene rhodium chloride dimer, cyclooctadiene iridium chloride dimer, and Fe(COD)(CO)3, and Crabtree's catalyst.
The M(COD)2 complexes with nickel, palladium, and platinum have tetrahedral geometry, whereas [M(COD)2]+ complexes of rhodium and iridium are square planar.
(E,E)-COD
 E,E-COD synthesis (Stöckmann et al. 2011)
E,E-COD synthesis (Stöckmann et al. 2011)
The highly strained trans,trans isomer of 1,5-cyclooctadiene is a known compound. (E,E)-COD was first synthesized by Whitesides and Cope in 1969 by photoisomerization of the cis,cis compound.[10] Another synthesis (double elimination reaction from a cyclooctane ring) was reported by Huisgen in 1987.[11] The molecular conformation of (E,E)-COD is twisted rather than chair-like. The compound has been investigated as a click chemistry mediator.[12]
References
- ↑ "AC1L1QCE - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Identification and Related Records. Retrieved 14 October 2011.
- ↑ Buehler, C.; Pearson, D. (1970). Survey of Organic Syntheses. New York: Wiley-Interscience.
- ↑ Shriver, D.; Atkins, P. (1999). Inorganic Chemistry. New York: W. H. Freeman and Co.
- ↑ Schiffer, Thomas; Oenbrink, Georg (2005), "Cyclododecatriene, Cyclooctadiene, and 4-Vinylcyclohexene", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a08_205.pub2
- ↑ Soderquist, John A.; Negron, Alvin (1998). "9-Borabicyclo[3.3.1]nonane Dimer". Org. Synth.; Coll. Vol., 9, p. 95
- ↑ Bishop, Roger. "9-Thiabicyclo[3.3.1]nonane-2,6-dione". Org. Synth.; Coll. Vol., 9, p. 692
- ↑ Díaz, David; Converso, Antonella; Sharpless, K. Barry; Finn, M. G. (2006). "2,6-Dichloro-9-thiabicyclo[3.3.1]nonane: Multigram Display of Azide and Cyanide Components on a Versatile Scaffold" (PDF). Molecules. 11 (4): 212–218. doi:10.3390/11040212.

- ↑ Schunn, R.; Ittel, S. (1990). "Bis(1,5-cyclooctadiene)nickel(0)". Inorg. Synth. 28: 94. doi:10.1002/9780470132593.ch25. ISBN 978-0-470-13259-3.
- ↑ Crascall, L; Spencer, J. (1990). "Olefin Complexes of Platinum". Inorg. Synth. 28: 126. doi:10.1002/9780470132593.ch34. ISBN 978-0-470-13259-3.
- ↑ Whitesides, George M.; Goe, Gerald L.; Cope, Arthur C. (1969). "Irradiation of cis,cis-1,5-cyclooctadiene in the presence of copper(I) chloride". J. Am. Chem. Soc. 91 (10): 2608–2616. doi:10.1021/ja01038a036.
- ↑ Boeckh, Dieter; Huisgen, Rolf; Noeth, Heinrich (1987). "Preparation and conformation of (E,E)-1,5-cyclooctadiene". J. Am. Chem. Soc. 109 (4): 1248–1249. doi:10.1021/ja00238a046.
- ↑ Stöckmann, Henning; Neves, André A.; Day, Henry A.; Stairs, Shaun; Brindle, Kevin M.; Leeper, Finian J. (2011). "(E,E)-1,5-Cyclooctadiene: a small and fast click-chemistry multitalent". Chem. Commun. doi:10.1039/C1CC12161H.

2.png)